Dioxygen (O2) is a potent little molecule that distinguishes Earth from other planets in the Solar System. The high oxygen content of the air we breathe is considered essential to sustain life as we know it, and yet it was not an original constituent of Earth’s atmosphere. Like other rocky planets in the Solar System, Earth’s initial atmosphere was mostly oxygen free. That is not to say that oxygen did not exist on the planet. It just means that the balance between processes that released or consumed oxygen was shifted towards oxygen consumption, so that only trace amounts of oxygen were left over from chemical reactions to be released back into the environment.
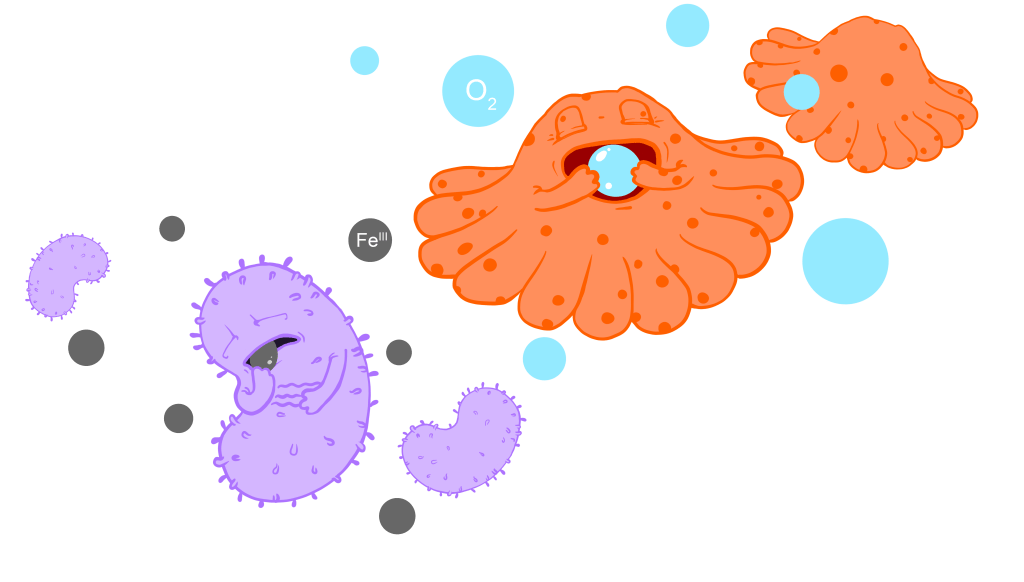
While today oxygen is primarily produced by green plants and phytoplankton, this was not the case on primordial (early) Earth. The first organisms to develop oxygen-releasing photosynthesis, the skill to convert solar energy into chemical energy by splitting water and releasing oxygen as a waste product, were cyanobacteria (figure 1). When exactly cyanobacteria developed the ability to do oxygen-releasing photosynthesis is poorly constrained, but geological evidence suggests that they became significant contributors of oxygen into the atmosphere by ~2.4 billion years ago (figure 1).
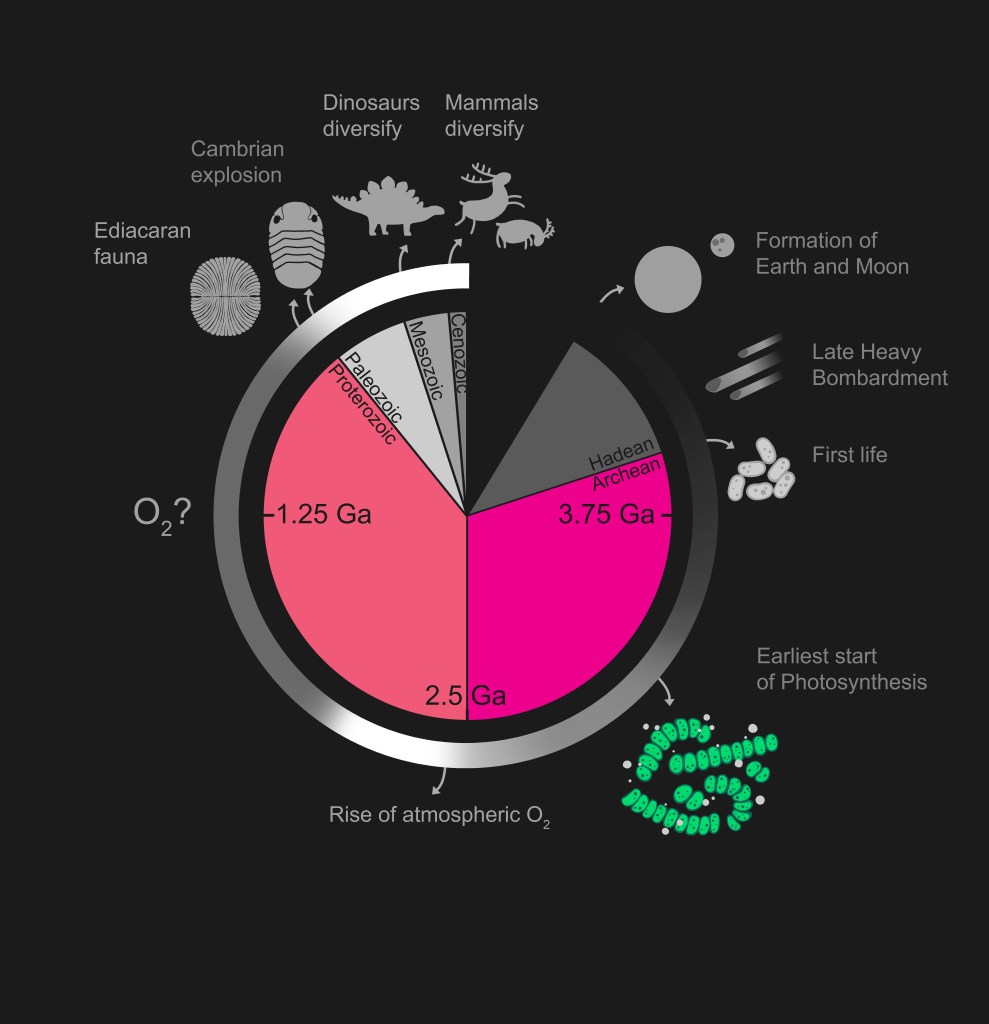
The time period over which oxygen became a persistent component of the atmosphere is known as the Great Oxidation Event (GOE). Several lines of evidence pinpoint the start of the GOE at ~2.4 billion years ago. Over the subsequent 400 million years, oxygen levels reached to at least 10% of the modern level (modern oxygen level in the atmosphere is ~21%). It is worth mentioning that, for the first half of Earth’s 4.6-billion-year history, primitive (early-stage) organisms were not concerned about breathing oxygen. On the contrary, they were happy to avoid it. Thus, the GOE likely wreaked havoc in food chains and brought significant rearrangements in ecosystems.
Microbes can thrive in various settings, including those where oxygen is not readily available. In oxygen-poor environments, microbes respire anaerobically (without oxygen) by consuming other ions (charged atom or molecule) or molecules, such as manganese oxides (MnO2), ferrous iron (Fe2+), nitrate (NO3–) or sulfate (SO42-). However, the energy gain from anaerobic respiration is insufficient to support the growth of complex multicellular organisms. Consequently, microbes that consume other molecules instead of oxygen can grow in numbers but not in cell size or complexity. Because the GOE should have removed the constraints on animal evolution posed by oxygen availability in their habitats, one might expect to see animal evolution take off, but this is not the case. Interestingly, the earliest animal fossils are found much later in the rock record than evidence for the GOE – so much later that dinosaurs could have evolved and become extinct ten times over!
The earliest animal fossils appear in the rock record by the Ediacaran period (635 – 541 million years ago) and diversify during the Cambrian period (541 – 485 million years ago), more than a billion years after oxygen first began to accumulate in the air. This discrepancy between animal evolution and Earth’s oxygenation has been puzzling scientists for decades. Various explanations have been put forward, but the exact processes leading to a modern-like atmosphere and the link to animal evolution remain subjects of debate and ongoing research. The rock record acts as a kind of time machine for geoscientists, helping them explore environments of the past.
This blog will introduce some of the popular ideas concerning the co-evolution of life and Earth’s evolving environments as well as how geologists conduct their research and recreate ancient, future, and alien worlds.
Text: Kärt Paiste (edits by Ichiko Sugiyama)
References
- Catling D.C., Glein C.R., Zahnle K.J., Mckay C.P., 2005. Why O2 is required by complex life on habitable planets and the concept of planetary ‘oxygenation time’. Astrobiology 5, 415–438.
- Farquhar, J., Zerkle, A.L., Bekker, A., 2014. Geologic and Geochemical Constraints on Earth’s Early Atmosphere, in: Treatise on Geochemistry. Elsevier, pp. 91–138.
- Holland, H.D., 2006. The oxygenation of the atmosphere and oceans. Philos T R Soc B 361, 903–915.
- Kasting, J., 1993. Earth’s early atmosphere. Science 259, 920–926

One thought on “Introduction”